Becton, Dickinson and Company BDX, popularly known as BD, recently announced the enrollment of the first patient in the investigational device exemption (IDE) study, AGILITY, which aims to evaluate the efficacy and safety of the BD Vascular Covered Stent in treating peripheral arterial disease (PAD).
The first patient in the AGILITY study was enrolled at Trinity Medical Center in Bettendorf, Iowa, by Dr. Nicolas Shammas, Interventional Cardiologist, Cardiovascular Medicine, PLLC.
Price Performance
For the past six months, BDX’s shares have lost 13.6% against the industry’s rise of 2.8%. The S&P 500 increased 13.4% in the same time frame.
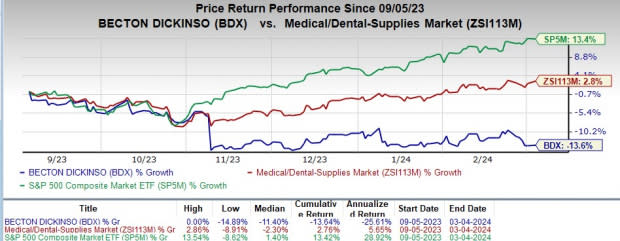
Image Source: Zacks Investment Research
More on the News
The Vascular Covered Stent, under investigation in the AGILITY study, is a low-profile, self-expanding nitinol implant enclosed in polytetrafluoroethylene. It is administered using a delivery mechanism that allows for regulated stent release.
The global, prospective, multi-center, single-arm, non-randomized AGILITY clinical research, according to BD, will involve up to 40 clinical study sites spread across the United States, Europe, Australia, and New Zealand, and 315 patients. Every treated patient will have follow-up at different intervals following therapy, ranging from one month to three years.
Per CDC, there are approximately 6.5 million people aged 40 years and older in the United States with PAD. Moreover, the global PAD patient population was 236 million per this article on National Library of Science. This population should have grown over the past years. It is a potentially severe illness that raises the risk of heart problems and amputation of a limb. Increased blood flow through the sick areas can be achieved with minimally invasive procedures using devices such as covered stents, drug-coated balloons, angioplasty balloons, and atherectomy.
Industry Prospects
Per a report by Market Research Future, the global peripheral artery disease market size was valued at $2.5 billion in 2022 and is expected to reach more than $4.9 billion by 2032 at a growth rate of 7.8%.
The market for peripheral artery diseases is expanding due to factors like the rapid increase in aging populations and the consequent rise in the prevalence of peripheral artery diseases, as well as an increase in product approvals.
With the given market potential for peripheral artery disease, BD’s Vascular Covered Stent can play a major role in the treatment of PAD and help boost the company’s revenues.
Notable Developments
In January 2024, BDX announced a strategic collaboration agreement with Techcyte. The tie-up aims to offer an artificial intelligence (AI)-based algorithm that guides cytologists and pathologists to identify evidence of cervical cancer and pre-cancer using whole-slide imaging efficiently and effectively.